Are We Ready for Post-Extubation Dysphagia?
By Karen Sheffler, MS, CCC-SLP, BCS-S of SwallowStudy.com
I have a secret! I have been wanting to write a definitive blog about post-extubation dysphagia for years. However, I have shied away from it, as there have been no clear answers for you. There is not one standard protocol or guideline to share for exactly when and how to evaluate this population of people who have just had their breathing tube removed after requiring intubation and ventilation for 48 hours or more. Guess what? Everyone is an individual. I can, though, provide a review and resources of post-extubation dysphagia, as well as some thoughts for the times we are in now in this COVID-19 pandemic. Right now, there are a lot of questions. We are #InThisTogether! Please use comment section below…

Dysphagia Evaluation & Management in a COVID-19 Era
March went out like a lion in the US.
When I started writing this on March 31st, there were 177,452 cases of COVID-19 in the USA, per the Johns Hopkins University of Medicine Coronavirus Resource Center. We are over 245,000 on 4/3/20, and we are only just starting to ramp up testing. We are all struggling to flatten the curve by staying home for weeks to months. Our healthcare heroes are there for us every day, even without the appropriate amount of personal protective equipment (PPE). Communities around the world so appreciate their sacrifices. There were chalk drawings around a Boston area hospital this week, with encouraging sayings like: “We lift each other up.” Everyone is yearning for the after, and therapists are collaborating to find best practices for rehabilitation after a person’s critical illness subsides.
Speech-language pathologists who work with people with difficulty swallowing (dysphagia) are ready! On social media, we have been already asking:
Will post-extubation dysphagia be significant, and will it go under-identified?
For well over 20 years, SLPs have known that the “removal of the endotracheal tube…does not automatically restore normal laryngeal functioning” for a normal swallow (Goldsmith, 2000, p 219). Will the whole medical team keep this in mind for all people with COVID-19 at the time of their extubation?
Per a national survey in 2012, only 41% of hospitals used a dysphagia screening protocol before starting oral intake post-extubation (Macht, et al., 2012). SLPs are worried about the increased risks for COVID patients to acquire an additional aspiration pneumonia, become re-intubated, and have prolonged hospitalizations (Leder & colleagues, 2019). Dr. Madison Macht, a critical care/pulmonary/neuro-critical care physician, who frequently writes about evaluating and treating dysphagia in the ICU, already noted in 2013 that,
“Post-extubation dysphagia is an under-recognized and potentially costly form of impairment in survivors of critical illness… (p8).”
A 2019 study by Patel and colleagues found that inpatients with dysphagia in the United States have a longer length of stay and a higher mortality than inpatients without dysphagia, and the study strongly advised:
“earlier recognition and intervention should be considered for all inpatients with dysphagia” to improve peoples’ outcomes and to reduce the overall costs and burden on healthcare.
General Intensive Care Considerations:
In addition to the risk for post-extubation dysphagia, will the many other intensive care factors worsen people’s swallowing safety and efficiency? Many of these issues will apply to any person with COVID with a prolonged ICU stay, even for those who did not require mechanical ventilation. Examples of these related risk factors are:
- New prone positioning used for people with COVID-19. Will this increase aspiration risk and decrease access to thorough oral care? (Read more about prone position guidelines here: DoD COVID-19 Practice Management Guide),
- Significant and prolonged respiratory compromise, causing difficulty coordinating breathing and swallowing to protect the airway. (This is an issue even if a person did not require intubation/ventilation. If the person is working hard at breathing with a respiratory rate over 25-30 breaths per minute, they may have a hard time squeezing in the swallow, as we cannot breath and swallow at the same time – making eating/drinking more dangerous. See a brief discussion of respiratory rate in this blog.)
- Prolonged length of stay in intensive care unit (ICU).
- ICU delirium and cognitive impairments.
- ICU myopathy/polyneuropathy (muscle weakness/paresis in people who are critically ill) with people with COVID-19 who do not always respond well to typical early mobilization efforts and have severe deconditioning.
- Additional suspected neurological manifestations by the COVID-19 virus
- Lethargy/sedation,
- Medication side-effects,
- Potential lack of thorough oral care / oral infection control practices,
- History of COPD, advanced liver disease, and sepsis were associated with aspiration and silent aspiration (see Marvin, et al., 2019). See also Stevie Marvin’s 2015 discussion of post-extubation dysphagia on Dysphagia Cafe.
- That list does not even mention the impact to quality of life and the mental health factors related to isolation, stress, and critical illness (we know that stress and critical illness can alter the normal healthy oral flora as well).
Post-Extubation Dysphagia Details:
Post-extubation dysphagia is an under-identified iatrogenic issue posing risks for serious medical complications (Brodsky, et al., 2018). These complications may be preventable with early identification.
Iatrogenic dysphagia = difficulty swallowing safely and efficiently caused by a medical treatment or treatments, such as prolonged intubation and/or traumatic intubation.
We know that people with COVID-19 have required prolonged intubations for mechanical ventilation. On March 30, 2020, Bhatraju, et al. (2020) reported that on 24 critically ill people in the Seattle area, showing that 75%* needed mechanical ventilation. The earliest that an individual was extubated (removal of the breathing tube) was 8 days. (We have heard anecdotal reports of up people requiring up to 14 days). Bhatraju commented on how a typical indicator of age did not seem to matter, as the age range of those intubated was 18 to 88 years.
*From early studies in China, overall, about 15% of people will become seriously ill and 5% of people will require the ICU, per a review in the DoD COVID-19 Practice Management Guide.
In post-extubation dysphagia research, El Solh and team (2003) noted that older age was a significant factor in which individuals had aspiration post-extubation. Age was an independent predictor of dysphagia in the 2014 Skoretz and colleagues’ post-extubation study of cardiovascular surgical patients. A multivariable analysis in the Marvin, et al. (2018) study showed that age (>65) was significantly associated with SILENT aspiration. We must keep in mind that age may or may not be a risk factor for post-extubation dysphagia during the COVID recovery process. Do we need to be more cautious regarding the risk for silent aspiration in those with COVID over 65? It is something to consider and start tracking, but without instrumental evaluations, how do we know for sure?
Why Do We Worry About Prolonged Intubation?
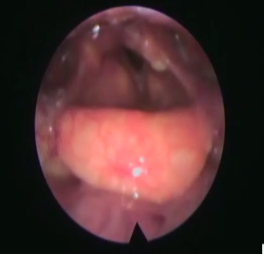
Image from Fiberoptic Endoscopic Evaluation of Swallowing. Laryngeal asymmetries after a prolonged intubation, which lead to post-extubation dysphagia.
- Pharyngeal, laryngeal & tracheal pathologies: (List per: Postma, et al., 2007, studying Fiberoptic Endoscopic Evaluation of Swallowing/FEES images of intubated patients with mean duration of 13 days. List below also per Brodsky, et al., 2018, who noted that injury was more often in those who were intubated for 5-10 days versus less than 5 days.)
- Edema (swelling) of inter-arytenoid space or more diffuse edema
- Erythema
- Granuloma/granulation tissue
- Vocal fold paresis/immobility
- Mucosal lesions
- Vocal fold bowing
- Airway/glottic stenosis
- Subglottic stenosis
- Subglottic mucosa edema
- Arytenoid(s) dislocation
- Ulcerations
- Secretions & other debris dried onto pharyngeal and laryngeal cavities must be added to this list, as this can make oral intake initially impossible without cleaning and humidification (per Rebecca Scheel’s videos presented at the December 2016, Dysphagia Grand Rounds in Boston after the publication of Scheel, et al., 2016).
- Significant changes in motor function and sensory input to the pharynx and larynx (Macht, et al., 2013). Altered chemo- and mechano-receptors in the pharynx and larynx (de Larminat, et al., 1995). Weakness and lack of sensation are easy to imagine. The individual who is intubated for 2-14 days will be atrophied from not using the swallowing mechanism to eat, drink or even swallow the quart of saliva we process every day. Use it or lose it is a common neuroplasticity principle.
- Delayed pharyngeal swallow response: Some descriptive studies show that even when participants with prior neurological deficits are excluded, people have a delayed pharyngeal swallow response after extubation. Researchers (V. de Larminat, et al., 1995) injected saline water into the pharynx on the day of extubation, and 62% of participants had significant delays in swallow response time. Delays were still present but improved at day 1 and day 2, but the pharyngeal swallow trigger returned to normal at 7 days post. Note: Brodsky, Pandian & Needham (2020) commented on how this was research with many limitations. Interestingly, though, Rebecca Scheel, et al. (2016) also noted that a pharyngeal swallow response delay was the most common descriptive finding with FEES testing.
What Happens to the Safety & Efficiency of the Swallow Post-Extubation?
- Brodsky, et al. (2018) found the following post-extubation symptoms:
- Dysphonia: 76% (Note: along with hoarseness and vocal cord impairment/hypomobility, we suspect reduced sensation of airway invasion, as well as a weak cough to eject the material.)
- Pain (odynophagia means pain with swallow): 76%
- Dysphagia: 49%
- Laryngeal dyspnea: 23%
- Stridor: 7%
- Post-extubation dysphagia & Intubation durations: Dysphagia can cause safety issues (causing aspiration and poor airway protection) and efficiency concerns (leaving significant residue stuck in the mouth, pharynx, and esophagus that can get into the larynx and airway after the swallow). Per a systematic review by Skoretz and colleagues in 2010, the incidence of dysphagia ranged from 3% to 62%. Of course, no study design was the same! Only 14 studies meet inclusion criteria. The studies that showed the highest incidence of dysphagia contained heterogeneous patients with the most prolonged intubations (up to 14 days). Sound familiar to COVID?
Skoretz, et al. (2014) analyzed 909 people who had cardiovascular surgeries, grouping them into 4 intubation-duration groups. Of those with intubations longer than 48 hours, 67.5% had dysphagia. (Compare that with the three other groups: <12 hours = 1%, 12-24 hours = 8%, and 24-48 hours = 16.6%).
More Research on Duration of Intubation: it is tough to make a blanket statement that duration of intubation is a perfect predictor of dysphagia. Leder, et al. (2019) found that an intubation duration of 4 days or more showed an increased incidence of dysphagia. Macht, et al. (2013) studied neurologically impaired people and found an independent association between intubation for >7 days and moderate-severe dysphagia. (Additionally, 66% of those with moderate-severe dysphagia had persistent difficulty swallowing, increased length of stay, and need for feeding tubes). Brodsky, et al. (2018) concluded:
“Evidence strongly indicates that intubation duration is associated with prevalence and severity of laryngeal injuries (p7).”
However, Marvin, et al. (2019) did not show an association between aspiration/silent aspiration and the duration of intubation. Scheel and colleagues at the Boston Medical Center (2016) reported on 59 participants who received a FEES 24-48 hours after extubation. (Participants had intubation durations of 2-28 days). Per her talk at our Boston Dysphagia Grand Rounds (December 13, 2016), she said they were surprised to find that the duration of intubation and the amount of time after intubation were NOT associated with dysphagia or predictors of aspiration. Descriptive data showed that “most” of the patients had dysphagia and “almost all” had laryngeal pathology, such as edema. The most common finding was a delayed pharyngeal swallow response. She noted that “25% of our patients aspirated, which is high for our small numbers.” She indicated that “emergent intubation” approached significance. I think this could be a key factor to look for in our COVID patients.
I still would advocate for caution with people who are COVID positive, especially those who required intubation for 48 hours or longer. I would keep in mind that at least half of those patients could have difficulty returning to normal oral intake (i.e., 51% incidence noted in Barker, et al., 2009).
- Swallow Safety / Aspiration: Studies vary widely with 25-68% of people aspirating when intubated longer than 48 hours. (Ajemain, et al., 2001, El Solh, et al., 2003, Scheel, et al., 2016; Hafner, et al., 2008; Marvin, et al., 2019). The incidence of silent aspiration (no obvious/overt signs to the nurse while material is going into the lungs) has ranged from 17.3% to 25% (Ajemain, et al., 2001; Leder, et al., 1998; Hafner, et al., 2008). Even more significant, Marvin and colleagues (2019) found silent aspiration on at least one consistency 56% of the time. Age was not associated with aspiration in general, but age was a significant factor in whether or not the aspiration was silent (Marvin, et al., 2019). Will nurses hear the weak throat clears and wet-gurgly voices through all the protective gear?
- Swallow Efficiency / Residue: Please let me know if anyone has research specifically on post-swallow residues in these people with post-extubation dysphagia.
So, When & How Do We Evaluate for Post-Extubation Dysphagia?
You still had to ask that question, didn’t you!?
My old rule-of-thumb had been (since starting in ICUs in 1999):
If the person was intubated for more than 48 hours, than it may take more than 24-48 hours to return the swallow to a more normal function. Caution should be used, and we look at many individual factors case-by-case.
This does not mean that I always waited a full 24 hours before initiating a swallowing evaluation, but my practice did include doing instrumental evaluations often – once the person was stable post-extubation. This rule-of-thumb may have come from Dr. Steven Leder’s 1998 study that recommended to evaluate for aspiration and dysphagia objectively “and/or delay oral intake for at least 24-48 hours to allow for optimal swallowing success” (p211, Leder, et al., 1998).
However, for people who are COVID-19 positive, we may not be able to use objective instrumental evaluations readily; more on that at the end of this section…
Leder continued to urge the field forward from the 1990’s. In 2016 at the Dysphagia Research Society meeting in Tucson, Leder initially presented his team’s now published work (Leder, et al., 2019). Click here for another blog on this topic. He advocated to not wait the typical 24 hours anymore*, saying that if the person was stable from an alertness and respiratory standpoint, they could return to oral intake faster via the valid Yale Swallow Protocol screening measure, which was confirmed in the study with FEES.
(*Note: patients were excluded in that study if they were not deemed stable by the medical team. A water swallow screen was not done on all patients post-extubation. So, screen your alert and stable patients, but take caution when patients are not deemed ready. Many people with COVID may not be immediately ready post-extubation.)
Prior to these COVID times, the SLPs at our Boston-area Dysphagia Grand Rounds noted that they were no longer waiting 24 hours “on purpose.” The general impression was that we take it on a case-by-case basis and do not have a standard policy to wait 24 hours. However, this concept of waiting 24 hours was still discussed in the newer Marvin, et al. (2019) study, and maybe it should be considered in these COVID times. That study showed significant improvements between the FEES done 2-4 hours after extubation versus the FEES performed after 24 hours (between 24-26 hours). While they were able to get 69% (47% on modified diet, 22% on regular) of participants on some kind of diet at 2-4 hours, they had more people on diets by 24-48 hours (76% were on diets, with 51% improving solid levels and 47% upgrading liquids). Their study may have suggested that if we wait 24 hours:
- maybe the diet recommendations would be less restrictive, AND we may
- reduce aspiration on a very early study,
- save staff hours,
- avoid over testing, and
- avoid patient disapproval of very modified diets and thickened liquids.
In these COVID times, the challenge for clinicians is the word “objectively.” Seasoned clinicians can make darn good clinical judgments based on their thorough chart reviews, lengthy discussions with medical team/family, and bedside swallowing evaluations that include cranial nerve and cough evaluations (we have to avoid testing cough now or perform it very subjectively with discussion with a nurse or listening while practicing physical distancing). But, bedside exams cannot objectively and completely evaluate the oropharyngeal swallow or rule-out aspiration. We document the limitations of our bedside evaluations; we document our judgements as “suspicions” and estimated levels of risk. This is what we have at this time. The objective instrumental measure of FEES certainly helped Scheel’s (2016), Marvin’s (2019) and Leder’s (2019) teams confidently get people on least restrictive diets quickly at rates of: 79%, 76%, and 82% of the time, respectively. BUT, now, with people who are positive with COVID-19, there may be no immediate FEES or bringing individuals to the radiology suite for videofluoroscopic evaluations of swallowing/VFSS. See the American Academy of Otolaryngology-Head and Neck Surgery and CMS Adult Elective Surgery and Procedures Recommendations:
- https://www.entnet.org/content/academy-supports-cms-offers-specific-nasal-policy-1
- https://www.cms.gov/files/document/31820-cms-adult-elective-surgery-and-procedures-recommendations.pdf
SLP Perspective from the Front Lines in New York City:
Dr Luis Riquelme, PhD, CCC-SLP, BCS-S (Director of the Center for Swallowing & Speech-Language Pathology at New York Presbyterian Brooklyn Methodist Hospital, Brooklyn, New York) shared his SLP COVID-19 experience in an NFOSD sponsored video on March 31, 2020. He noted how this is all new and “what we know about extubation, for example, doesn’t always play out here.” He noted that when SLPs typically evaluate people after they have been extubated, the individuals have many complex underlying diseases/disorders to take into consideration. That is still the case with some people with COVID, but there are also people who were very healthy prior to COVID but still required intubation/ventilation. Now, even these previously healthy adults still may have respiratory compromise after extubation, per Riquelme. It is our job to look at each person as an individual. Brodsky and colleagues (2014b) found that there were no set practice patterns developed yet to guide post-extubation dysphagia evaluations and timing. Macht and colleagues in 2012 also reported on post-extubation variability in evaluations, instrumentals, and treatment techniques. Similarly, Dr Riquelme said there are no patterns yet with COVID patients; therefore, he advised to look at each person’s respiratory status and stability to help guide a safer process back to oral intake and eventually the least restrictive diet.
Brodsky and colleagues confirmed the importance of this case-by-case readiness approach in a 2020 pre-COVID summary article, which also stressed the importance of a multidisciplinary approach:
“…patients’ clinical readiness should be a marker for screening/evaluation, not a fixed duration of time post-extubation.”
The multidisciplinary approach includes trained staff performing a SCREENING of the swallow, using an approved valid screening protocol (such as the Yale Swallow Protocol). It also includes a “cautious approach” that entails having staff monitor all oral intake closely when it is first re-started for anyone who was recently extubated, may be lethargic/confused, or may have difficulty coordinating breathing and swallowing due to persistent difficulty breathing (Brodsky, et al., 2020, p94). Post-COVID, Brodsky commented that he is concerned about the increased durations of intubation and the frequent use of prone positioning, which could both contribute to increased laryngeal injury. These issues that will be in the forefront of research at the Johns Hopkins now (personal communication, 3/31/20).
Similarly, Dr Riquelme also described a multidisciplinary/teamwork method at the hospital in NYC. They are doing direct evaluations (SLP in the room) and indirect evaluations (where the SLP works with the RN who is going in the COVID room anyway). He stressed the importance of communication across all disciplines to increase patient safety, as well as frequent follow-ups checking for patient readiness. He noted that they document thoroughly, especially the rationales for why exam was performed indirectly, as well as documenting all the communications and collaborations with the medical team. SLPs can provide essential-life-saving recommendations (even when exam is indirect and collaborative only). Without the instrumental evaluations to make more confident and definitive recommendations, Riquelme advised:
- thorough chart reviews, including review of baseline and prior dysphagia and medications that may cause dysphagia,
- discussions with team/family,
- collaborating with nursing, and
- good infection control practices to minimize an additional aspiration pneumonia (i.e., thorough oral cleaning with teeth brushing for a thorough oral decontamination to prevent aspiration of oral pathogens).
Dysphagia Evaluations Without Instrumentals
SLPs will be relying on clinical judgement, critical thinking and communication skills with the patient and entire medical team. Our evaluations may look different. (For example, telepractice for your bedside dysphagia evaluations could make that indirect approach described by Dr. Riquelme a lot more efficacious and billable. Telepractice would minimize the need for an additional staff member doning PPE and entering a COVID room. Please see Dr. Georgia Malandraki’s Telepractice resources on her lab’s website at: https://www.purdue.edu/i-eatlab/telehealth-recommendations-for-dysphagia-management-during-covid-19/).
Per research by Stevie Marvin and colleagues (2019), we should not rely solely on the water swallow screen, which challenges continuous drinking of 90ml of water without stopping (part of most swallow screening measures and Yale Swallow Protocol). In the study by Marvin and colleagues (2019), silent aspiration occurred 36% of the time in the 53 participants who could complete a 90ml thin liquid water challenge. This may put into question the prior study by Leder and colleagues (2011) that suggested that “silent aspiration was volume-dependent.” Speech-language pathologists consider many more issues than just a positive cough/throat clear on a screening tool. SLPs are essential members of the COVID team to prevent complications from post-extubation dysphagia, especially due to:
- Prolonged intubations;
- Potential emergent and traumatic intubations;
- Significant respiratory distress persisting after extubation (or even in patients who have avoided the need for intubation) – causing difficulty coordinating breathing and swallowing;
- Prone positioning;
- Potential CNS involvement;
- Massive deconditioning (i.e., generalized weakness), as newer techniques of early mobilization have not been as beneficial in this group of people.
Bottom-line now:
We are dealing with a whole new medical crisis that has significant variability,
in this area of post-extubation dysphagia that has not been definitive,
with no standardized protocols or instrumental evaluations to guide us clearly through the fog!
But,
We are #InThisTogether. Please share in comments below!
Additionally, we can advocate for patients by reminding:
Don’t give the person who had prolonged intubation a glass of water to chug
the minute they get extubated
when the mouth and teeth have not been cleaned thoroughly in days!
Last thought,
There are some reports of cranial nerve and central nervous symptom (CNS) findings with this COVID-19 virus, so continue to perform thorough cranial nerve evaluations. See the American Academy of Otolaryngology – Head and Neck Surgery’s discussion of anosmia/hyposmia (lack of or decreased smell) and dysgeusia (lack of taste) and how to report it. Medical discussions on twitter revealed a finding of cranial nerve 5 involvement with trigeminal neuralgia on 3/24/20 by @MikeAlbertMD. Please also see this June 2020 research and Interview with Fabio Ferreli, MD, and Giuseppe Mercante, MD, authors of Prevalence of Taste and Smell Dysfunction.
We need to continue to share findings and lift each other up — contributing in any way we can, even if that means staying at home and researching for others, while your son jumps on the sofa and your husband practices clarinet scales! Find humor and music to stay sane, and
Thank you all!
*******
Post-Extubation Dysphagia & COVID-19 References & Resources:
COVID-19 Updates:
ASHA.org: https://www.asha.org/About/Coronavirus-Updates/
Johns Hopkins University Corona Virus Resource Center: https://coronavirus.jhu.edu
New articles on ICU & Post-Extubation Dysphagia (updated 5/26/20):
1. McGrath, B.A., Wallace, S. and Goswamy, J. (2020), Laryngeal oedema associated with COVID ‐19 complicating airway management. Anaesthesia. doi:10.1111/anae.15092
https://onlinelibrary.wiley.com/doi/10.1111/anae.15092
McGrath & colleagues stated: “during this pandemic, we encourage colleagues to prepare for difficulties during tracheal intubation and extubation, to be vigilant and actively look for post‐extubation dysphagia and dysphonia, and to seek early advice from colleagues in head and neck surgery and in speech and language therapy.”
2. Brodsky, Martin B. PhD, ScM, CCC-SLP1,2; Nollet, Joeke L. MD, MSc3; Spronk, Peter E. MD, PhD4; González-Fernández, Marlís MD, PhD1 (2020). Prevalence, Pathophysiology, Diagnostic Modalities and Treatment Options for Dysphagia in Critically Ill Patients. American Journal of Physical Medicine & Rehabilitation: April 16, 2020 – Volume Publish Ahead of Print – Issue – doi: 10.1097/PHM.0000000000001440 LINK
More Webinars, Blogs & Podcasts: (Updated 4/27/20)
Swallow Your Pride: At the Table with Martin Brodsky & Paula Leslie: A Conversation on COVID https://www.mobiledysphagiadiagnostics.com/132-at-the-table-with-martin-brodsky-paula-leslie-a-conversation-on-covid19/
See also the April 2020 Swallow Your Pride Podcast with Jacqueline Connell, MS, CCC-SLP from NYC.
References:
Ajemian, M.S., Nirmul, G.B., Anderson, M.T., Zirlen, D.M. & Kwasnik, E.M. (2001). Routine Fiberoptic Endoscopic Evaluation of Swallowing Following Prolonged Intubation: Implications for Management. Arch Surg., 136(4), 434–437. doi:10.1001/archsurg.136.4.434 https://www.ncbi.nlm.nih.gov/pubmed/11296115
Barker, J., Martino, R., Reichardt, B., Hickey, E. J., & Ralph-Edwards, A. (2009). Incidence and impact of dysphagia in patients receiving prolonged endotracheal intubation after cardiac surgery. Canadian journal of surgery. Journal Canadien de Chirurgie, 52(2), 119–124. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2663495/
Brodsky, M. B., Gellar, J. E., Dinglas, V. D., Colantuoni, E., Mendez-Tellez, P. A., Shanholtz, C., . . . Needham, D. M. (2014a). Duration of oral endotracheal intubation is associated with dysphagia symptoms in acute lung injury patients. Journal of Critical Care, 29, 574–579. https://doi.org/10.1016/j.jcrc.2014.02.015
Brodsky, M. B., González-Fernández, M., Mendez-Tellez, P. A., Shanholtz, C., Palmer, J. B., & Needham, D. M. (2014b). Factors associated with swallowing assessment after oral endotracheal intubation and mechanical ventilation for acute lung injury. Annals of the American Thoracic Society, 11(10), 1545–1552. https://doi.org/10.1513/AnnalsATS.201406-274OC
Brodsky, M.B., Levy, M.J., Jedlanek, E., Pandian, V., Blackford, B., Price, C., et al. (2018). Laryngeal injury and upper airway symptoms after oral endotracheal intubation with mechanical ventilation during critical care: A systematic review. Critical Care Medicine, DOI: 10.1097/CCM.0000000000003368 https://www.ncbi.nlm.nih.gov/pubmed/30096101
Brodsky, M.B., Pandian, V. & Needham, D.M. (2020). Post-extubation dysphagia: a problem needing multidisciplinary efforts. Intensive Care Med, 46, 93–96. https://doi.org/10.1007/s00134-019-05865-x
Coyle, J. L. (2014). Dysphagia Following Prolonged Endotracheal Intubation: Is There A Rule of Thumb?. Perspectives on Swallowing and Swallowing Disorders (Dysphagia), 23(2), 80-86. LINK
de Larminat, V., Montravers, P., Dureuil, B. & Desmonts, J.M. (1995). Alteration in swallowing reflex after extubation in intensive care unit patients. Critical Care Medicine, 23(3), 486-490.https://journals.lww.com/ccmjournal/pages/articleviewer.aspx?year=1995&issue=03000&article=00012&type=abstract
El Solh, A., Okada, M., Bhat, A., Pietrantoni, C. (2003). Swallowing disorders post orotracheal intubation in the elderly. Intensive Care Med., 29(9), 1451–1455. DOI: 10.1007/s00134-003-1870-4 https://www.ncbi.nlm.nih.gov/pubmed/12904855
Goldsmith T. (2000). Evaluation and treatment of swallowing disorders following endotracheal intubation and tracheostomy. Int Anesthesiol Clin., 38(3), 219–242. https://www.ncbi.nlm.nih.gov/pubmed/10984854
Hafner, G., Neuhuber, A., Hirtenfelder, S., Schmedler, B., & Eckel, H. E. (2008). Fiberoptic endoscopic evaluation of swallowing in intensive care unit patients. European archives of otorhinolaryngology: Official journal of the European Federation of Oto-Rhino-Laryngological Societies (EUFOS): Affiliated with the German Society for Oto-Rhino-Laryngology – Head and Neck Surgery, 265(4), 441–446. https://doi.org/10.1007/s00405-007-0507-6
Kwok, A. M., Davis, J. W., Cagle, K. M., Sue, L. P., & Kaups, K. L. (2013). Post-extubation dysphagia in trauma patients: It’s hard to swallow. The American Journal of Surgery, 206, 924–927; discussion 927–928. https://doi.org/10.1016/j.amjsurg.2013.08.010
Leder, S. B., Cohn, S. M., & Moller, B. A. (1998). Fiberoptic endoscopic documentation of the high incidence of aspiration following extubation in critically ill trauma patients. Dysphagia, 13, 208–212.https://www.ncbi.nlm.nih.gov/pubmed/9716751
Leder, S.B., Suiter, D.M. & Green BG. (2011). Silent aspiration risk is volume- dependent. Dysphagia, 26,304–9. https://link.springer.com/article/10.1007/s00455-010-9312-2
Leder, S. B., Warner, H. L., Suiter, D. M., Young, N. O., Bhattacharya, B., Siner, J. M., … Schuster, K. M. (2019). Evaluation of Swallow Function Post-Extubation: Is It Necessary to Wait 24 Hours? Annals of Otology, Rhinology & Laryngology, 128(7), 619–624. https://doi.org/10.1177/0003489419836115 https://www.ncbi.nlm.nih.gov/pubmed/30841709
Macht, M., Wimbish, T., Clark, B. J., Benson, A. B., Burnham, E. L., Williams, A., & Moss, M. (2012). Diagnosis and treatment of post-extubation dysphagia: results from a national survey. Journal of critical care, 27(6), 578–586. https://doi.org/10.1016/j.jcrc.2012.07.016
Macht, M., King, C. J., Wimbish, T., Clark, B. J., Benson, A. B., Burnham, E. L., Williams, A., & Moss, M. (2013). Post-extubation dysphagia is associated with longer hospitalization in survivors of critical illness with neurologic impairment. Critical care (London, England), 17(3), R119. https://doi.org/10.1186/cc12791
Macht, M. (2013, October). Dysphagia and swallowing disorders in the ICU (Review). Retrieved from PulmCCM https://pulmccm.org/review-articles/dysphagia-swallowing-disorders-icu-review/
Marvin, S. (2015, April). Managing post-extubation dysphagia. Retrieved from Dysphagia Café https://dysphagiacafe.com/2015/04/16/post-extubation-dysphagia-aspiration-pneumonia-fees/
Marvin, S., Thibeault, S. & Ehlenbach, W.J. Post-extubation Dysphagia: Does Timing of Evaluation Matter? Dysphagia, 34, 210–219 (2019). https://doi.org/10.1007/s00455-018-9926-3
Patel, D. A., Krishnaswami, S., Steger, E., Conover, E., Vaezi, M. F., Ciucci, M. R., & Francis, D. O. (2018). Economic and survival burden of dysphagia among inpatients in the United States. Diseases of the esophagus: Official journal of the International Society for Diseases of the Esophagus, 31(1), 1–7. https://doi.org/10.1093/dote/dox131
Postma, G.N., McGuirt, W.F., Sr, Butler, S.G., Rees, C.J., Crandall, H.L. and Tansavatdi, K. (2007). Laryngopharyngeal abnormalities in hospitalized patients with dysphagia. The Laryngoscope, 117, 1720-1722. doi:10.1097/MLG.0b013e31811ff906 https://www.ncbi.nlm.nih.gov/pubmed/17690603
Rassameehiran, S., Klomjit, S., Mankongpaisarnrung, C., & Rakvit, A. (2015). Postextubation Dysphagia. Proceedings (Baylor University. Medical Center), 28(1), 18–20. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4264700/
Scheel, R., Pisegna, J. M., McNally, E., Noordzij, J. P., & Langmore, S. E. (2016). Endoscopic Assessment of Swallowing After Prolonged Intubation in the ICU Setting. Annals of Otology, Rhinology & Laryngology, 125(1), 43–52. https://doi.org/10.1177/0003489415596755 https://www.ncbi.nlm.nih.gov/pubmed/26215724
Skoretz, S. A., Flowers, H. L., & Martino, R. (2010). The incidence of dysphagia following endotracheal intubation: A systematic review. Chest, 137, 665–673. https://www.ncbi.nlm.nih.gov/pubmed/20202948
Skoretz, S. A., Yau, T. M., Ivanov, J., Granton, J. T., & Martino, R. (2014). Dysphagia and associated risk factors following extubation in cardiovascular surgical patients. Dysphagia, 29, 647–654. https://doi.org/10.1007/s00455-014-9555-4


God bless you for sharing your knowledge and experience. Here in Portugal, I work in a central hospital dealing with Covid and dysphagia, the context is an uncharted territory, and yours is a wonderful map, showing us how to proceed. Thank you, we are #InThisTogether
Good news for SLPs in Massachusetts, regarding telepractice – easing up of restrictions during this pandemic:
Dear Massachusetts ASHA Member:
The Massachusetts Board of Registration for Speech-Language Pathology and Audiology voted to approve a temporary policy that will remain in effect for the duration of the state of emergency.
The policy makes the following changes:
waives the in-person evaluation requirement for telepractice;
eliminates the need for training in advance of delivering telepractice services with the condition that the licensee obtain the 10 hours of training within four months of the start of telepractice;
extends any license or certificate issued by the Board which, during the state of emergency, has expired or will expire but is otherwise in good standing, until 90 days following the termination of the state of emergency; and
extends the timeframe to complete continuing education for renewal of licenses that expired or will expire during the state of emergency until 90 days following the termination of the state of emergency.
Questions?
Contact Susan Adams, ASHA’s director of state legislative & regulatory affairs, at sadams@asha.org.
An SLP’s suggestions for discussing COVID rule-out/positive patients with doctors when a swallow evaluation order is received.
“Here are some tips on how I’ve been framing the conversations that have worked:
· “I see you consulted for a swallow evaluation on patient XXX, who is being ruled out for COVID-19/has tested positive for COVID-19. We have written guidelines asking that all consult services think critically about limiting patient contact, when safely able, to help mitigate the risk of unnecessary exposure and spread of COVID-19, as well as to preserve PPE for essential care tasks. Our service is at a particular risk of becoming vectors given that we elicit coughing, are in close contact with the mouth/nose, cover all floors and units, and work with some of the most severely respiratory compromised/complex airway patients. Therefore, our service has been conducting thorough risk/benefit analyses on all consults in discussion with medical teams to ensure that we are meeting this hospital-wide directive. I’d love to take a moment now to discuss what concerns you had that led you to write the consult…”
Other points that might be appropriate depending on the patient’s status:
· We spoke to the patient’s family/facility and learned that the patient’s baseline is XXX and their baseline diet has been XXX. If there are no new problems, I would recommend continuing on that diet until testing is completed.
· In the differential right now, it seems that COVID-19 is a real concern, so once that is ruled out, we are more than happy to evaluate for aspiration as another potential etiology of current respiratory compromise.
· I notice that the CXR that you completed shows multiple bilateral opacities, and do you think that is more consistent with COVID-19 than with aspiration?
· I understand that one strategy the medicine teams are using to combat COVID-19 is putting patients on restrictive fluid intake, so holding NPO until the test results come back may not be a bad idea and might actually be helpful.
· The RN tells me that the patient is coughing with everything, so holding them NPO may be the best option, since COVID-19 patients are unable to travel to radiology for a video swallow study or receive a FEES.
· Where do you feel the patient is in their COVID course? Do you think they are on the mend or are they at risk of decompensating? If decompensation is still a risk, it is worth deferring evaluation and holding them NPO until we are sure they are stabilized.
· Given tenuous respiratory status (ask about respiratory rate – is it consistently over 25? Do they still require NIV, high flow or face mask?), placing temporary alternative means to wait and see the trajectory of their disease over the next few days/weeks seems most appropriate and fitting with hospital guidelines.
Other things we can offer, when we do not agree that an in-person visit is necessary:
· I’d be more than happy to complete a video consult with you, the patient, and/or the RN.
· I’d be happy to provide the RN with 2-3 strategies to try out to see if that addresses the concerns while we wait for these test results.
We also provide augmentative communication measures and aphasia suggestions for better receptive/expressive communication with healthcare staff.
There will be patients who are still being ruled-out or are POSITIVE that we will NEED to see, but I think we can use the above conversations to make sure that everyone is thinking critically about why/why not to see each patient. In particular, I think we need to help the doctors think about when during the disease process our input is beneficial/meaningful.”
I appreciate the deep reflections that you share in relation to this very delicate and complex matter. I request your authorization to translate this post and share with the speech therapist community in Spain, contributing in an important way to guide our professional work in these times of Covid-19. With my best wishes, take good care of yourselves!
Por Supuesto! Es muy importante para compartir esta información! Diga me si tienes preguntas. Ingles o Español – Karen
This is a great article. Often icu will not tell y’all about what happens after extubation. My mother just got extubated but is experiencing severe sore throat and unable to speak. She hasn’t eaten for days and didn’t pass the swallow test. They end up feeding her through ng tube. Can you offer some advice on how to regain swallowing and speech while recovering in the hospital. What are some exercises patient can do on there own to speed up throat and speech recovery.
First of, everyone is different, and often the amount of dysphagia (difficulty swallowing) and hoarseness depends on how long the person was intubated, how many times extubated/re-intubated, how traumatic the intubation was in the first place, and whether there was additional trauma to the voice box/vocal cords during intubation (e.g., self-extubation attempts or irregular positioning and movement with the tube through the vocal cords). We are just starting to see and question about the needed prone positioning during COVID-19 intubations. How is the head positioning? Is the head repeatedly turned to avoid skin breakdown? Is the head in extension – putting more pressure on the vocal cords? The voice will be just a whisper and/or very hoarse for days. We have to also check to see if the person is managing secretions –> does the voice sound wet/gurgly with pooled saliva? Sometimes so much swelling and obstruction and pain that the saliva does not swallow down.
Second, make sure your mom’s oral cavity is very clean. Brush teeth and scrub with mouthwash. The hospital should have a suction toothbrush and suction swabs to make this thorough. Pay special attention to scrubbing teeth, palate, gums. When someone is not eating/drinking for a long time, there can be a lot of build up of crud (for lack of a better word – and to not use a medical term like “biofilm”). This can contain a lot of bacteria. If the saliva is aspirated (goes down the wrong way when trying to swallow), then poor oral hygiene can really increase the person’s risk for developing an additional aspiration pneumonia (on top of the COVID19 pneumonia they are recovering from).
Third, re exercises and voice/swallow eval/treat: The best exercise for swallowing is swallowing! However, we have to do it safely. The mouth has to be clean (we call that oral decontamination or oral infection control). Maybe the speech pathologist (SLP) can try ice chips (one at a time), to stimulate the swallow. Before doing any PO trials and after the mouth is clean, the SLP does the first part of a bedside swallow evaluation: assessment of alertness, attention, orientation (aware of person, place, time), cognition, ability to express and comprehend language, voice quality, cough strength/productiveness, and a thorough oral sensory-motor exam. If the therapist finds that the person is too confused, lethargic or inattentive, they may not be safe for eating/drinking trials. If the therapist finds that the tongue and lips are very weak, then exercises can be targeted to specific strengthening of those areas. If the voice is very weak after extubation, the best method initially is to wait for healing. Sometimes we use the Ear Nose and Throat department to scope and see the vocal cords, but during COVID19, the ENT staff are minimizing the scoping due to high viral load. So we may have to wait it out and see if the vocal quality gets better. Make sure to have them use humidified shovel mask for humidified air for mouth/throat. This can clear up oral cavity and provide some necessary humidification for the damaged vocal cords. You don’t want to aggressively do vocal cord exercises without knowing what the vocal cords look like –> could make things worse.
Back to the swallow, If the person is safe for taking in some food/liquid, the therapist will proceed with caution. Typically, when we are concerned with a major difficulty swallowing and aspiration, we will do an instrumental evaluation (taking person to X-ray department for videoswallow study/VFSS or MBSS or dropping a scope through the nose to watch them eat/FEES). However, currently, most hospitals are not able to do that with COVID19. Therefore, bedside, we cannot rule-out aspiration. We cannot see if the liquid/food/saliva is going down the wrong way silently. Many people who are extubated will actually have silent aspiration — and we will not know bedside. No cough, throat clears, complaints. The person’s sensation is affected by the trauma and swelling. Sometimes initially, someone may be able to “tolerate” (we hope they are tolerating it) a few bites of applesauce (or a smooth/moist puree) and a thickened liquid. Note: thin or regular liquids drop down the wrong way fast. Thin liquid will be the hardest to control and swallow safely upon extubation – posing a very high risk for aspiration (and silent aspiration – the nurse will not know). What I have heard from many of my colleagues working in the ICU with people with COVID19 is TO GO VERY SLOWLY. Risk for reintubation is high. Risk for decline in function is high, especially due to weakness, respiratory instability, and the lethargy and confusion (delirium). Younger people are even having coughing or difficulty with thin liquid within the first few days, but they seem to be getting back to more regular diets in 1-2 weeks of healing time. Older individuals may take a lot longer. In addition to GO SLOW, the overall concept has been that of PATIENCE! It is a bit of a vicious cycle, as patience is needed in not taking someone off the vent (extubate) too quickly, so many are intubated/ventilated for over 10 days. That prolonged intubation in turn increases the dysphagia risk.
It is important to maintain good nutrition all throughout. People have tube feeding during intubation and often it needs to be continued after extubation for at least a few days. One colleague described that when the team pulls out the oral gastric tube (OG) during extubation, it then is then hard to reinsert the nasogastric tube (NG). Therefore, her hospital is starting to place the NGtube before extubation, so that they leave the NGtube in after removing the breathing tube and the OGtube. It is good your mom has the NGtube (that you mentioned), as adequate nutrition is essential for regaining muscle strength/healing. The registered dietitian nutritionist (RDN) will be watching nutrition/hydration requirements closely.
As your mom gets further out and at a rehab facility, the SLP and rehab team will take over with the strengthening necessary. I cannot suggest specific swallow exercises without knowing what are the specific appropriate targets for your mom. The SLP will try to target a program to her. One idea is when someone is able to swallow saliva and/or ice chips without pain, they could try to focus on an effortful swallow (feeling a push with the tongue and a squeeze with the throat – tuning into these muscle actions). Overall body restrengthening with OT/PT also helps tremendously with feeding/swallowing/voice recovery.
Hi! I wanted to share-
In managing COVID19 patients in a big teaching hospital in NYC (the epicenter), this article “Are we ready for Post-Extubation dysphagia (COVID19)” was a lead resource for us when we discussed post extubation considerations with our physicians. It also was a source of motivation to prompt us to really think critically and review data- what we may know and what we don’t
I also personally found your article “The Conversation and the SLP role in Palliative care” to be extremely helpful– as we are seeing many of these end of life cases now, my colleagues and I are developing new skills/ perspectives for managing these difficult patients and issues.
THANK YOU for your contributions to the field! I am always reading and listening!!
Thank you so much Talia. It has been great working with you this week now on the next issue of how to address the patients who did not extubate and need a trach placed. Next blog hopefully out tomorrow. I am so proud of our field and working together across healthcare systems and around the world!
Great information here. Thank you! My 19 yr old daughter was placed on a ventilator for 20 days battling covid and pneumonia. She was extubated on 4/24. We are now on day 5 post extubation, she is still very weak and cannot get out of bed and has not passed the swallow test. She’s been moved out of icu to step down where a speech therapist will meet with her for quick session. But so far I am told by nurses the sessions are not going well and they will “try again tomorrow”. My daughter had a feeding tube in her nose which she pulled out 2 days ago so they gave her iv with vitamins for nutrients until they can access her again. Do you have any suggestions? I haven’t seen my daughter since the day I dropped her off at the ER which was Apr 5th. When speaking to her, her voice sounds slow and soft and littler raspy. She has been saying more words each day when speaking but she gets frustrated and starts crying. Is there anything you suggest? Thank you
I am so sorry you are going through this with your young daughter. I hope people see how this can affect the young people too.
I hope you are able to talk directly with that speech pathologist about her/his findings. Hopefully she will tolerate at least a modified diet soon. Sometimes soft or blended foods and thickened liquids may be tolerated better than thin liquids. You could ask nurses to make sure they are helping her brush her teeth and using mouthwash well (3-4 times a day). If her oral hygiene is really good, you could ask the nurse, medical team and speech pathologist if she could have ice chips at least (after cleaning mouth). That way she would at least be practicing her swallow to get her swallow slowly better. Any pain in swallowing? The tricky thing is we are not able to scope with a camera to see the airway, vocal cords and throat to assess for damage, swelling, etc. That makes our evaluations less confident. We have to go slower and more carefully to prevent a decline in function or reintubation. I really hope you can talk with the speech pathologist for more direct ideas, as I cannot give detailed advice in this manner without evaluating her myself. Best wishes.