Dysphagia Digest 2017: Raising Awareness Regarding Swallowing Disorders & Treatment
There was not much time to sample Portland, Oregon’s craft beers or Portland’s famous Salt & Straw ice cream, because the over 500 attendees at the Dysphagia Research Society‘s annual meeting were in stimulating sessions from March 1-4, 2017. DRS is celebrating its 25th anniversary this year – 25 years of raising awareness about dysphagia. Give DRS a big “cheers” with your craft beer and read on!
The mayor of the city of Portland, Ted Wheeler, declared February 26th to March 4th to be Swallowing Disorders Awareness Week. He acknowledged the efforts of healthcare providers and researchers from all over the world who convened in Portland to combat the multifaceted problem of dysphagia through research and raising awareness.
If you have not followed this year’s annual meeting and post-graduate course on social media, then check out:
- Twitter: search with #DRS2017 or follow @Official_DRS, @SwallowStudySLP, @Kskrings, @rinkislp, @annacmiles, @justinroe, @hannekekalf, @MBBrodskyPhD, @MichelleCimoli, @IDDSI_org – just to name a few meeting tweeters!
- Facebook: @DysphagiaResearchSociety and @SwallowStudySLP
This DRS Digest will cover 5 Topics with Take-Home Messages.
These are not a top 5; they are just my first 5. I would really need hundreds of hours post-DRS to put together the extensive list of top findings!
1) The Oral Health Assessment Tool (OHAT):
Lee, Yan Shan (and colleagues: Yap, Xin Ying, Chia, An An & Wong, Seng Mun) presented on “Oral Health Screening by Speech and Language Pathologists (SLPs) Using the Oral Health Assessment Tool (OHAT).” The OHAT was modified by Chalmers, et al. in 2004 from the tool originally created by Kayser-Jones, et al. in 1995. Lee showed that with a brief training by a dental hygienist, SLPs can accurately and reliably screen for poor oral health status and make appropriate referrals for further dental intervention. SLPs were most accurate in rating salivary health, with accuracy extending to even the untrained SLPs.
Lee suggested the following informative articles on the OHAT:
- Chalmers, J.M., King, P.L., Spencer, A.J., Wright, F.A.C. & Carter, K.D. (2005). The Oral Health Assessment Tool–Validity and Reliability. Australian Dental Journal, 50 (3), 191-199. (This article contains the full OHAT chart.)
- Simpelaere, I.S., Gwen Van Nuffelen, G., Vanderwegen, J., Wouters, K. & De Bodt, M. (2016). Oral Health Screening: Feasibility and Reliability of the Oral Health Assessment Tool as Used by Speech Pathologists. International Dental Journal, 66 (3), 178-189.
Take Home Message: SLPs should receive training in how to identify unsafe oral conditions. SLPs can make a significant contribrution to the patient’s overall health by raising awareness of the importance of oral hygiene. The SLP can incorporate a structured oral health rating tool into every bedside/clinical swallowing evaluation. Maybe SLPs can begin to repair the split that occurred 150 years ago when the medical and dental professions parted ways. (Read more in this resource on Oral Care & Aspiration Pneumonia.)
2) New Zealand Secretion Scale (NZSS):
The predictive value of accumulated secretions.
Can the amount and patient’s ability to detect secretions predict aspiration and pneumonia risk? It is certainly important to describe and rate secretions when conducting Fiberoptic Endoscopic Evaluations of Swallowing (FEES). However, most secretion scales address only the location and amount of secretions, but the NZSS also takes patient’s response into account. Anna Miles and colleagues, from Auckland, New Zealand, have developed a 3-part scale with a maximum score of 7 out of 7 for the most severe pooling of secretions in pyriforms and laryngeal vestibule without any reaction to cough or clear them out.
Just in case you hear someone referring to this by it’s initials, you need to recall that a “z” in many countries is pronounced “zed!” Therefore, you will hear this scale referred to as the “NZedSS.” That threw me for a loop initially when listening to the terrific presentation by Anna Miles.
Miles and colleagues studied a large cohort of patients from acute care (“pretty sick patients,” per Miles, with 31% from critical care). The patients’ pneumonia rates on their admissions were significantly associated with the NZSS score, the Penetration/Aspiration Scale score, the presence of a tracheostomy, and having a cardiovascular etiology.
Update from February 2018: Here is their newest publication in the Dysphagia Journal, looking at the “Predictive value of the New Zealand Secretion Scale (NZSS) for Pneumonia.”
Update August 2017: Here is the publication in the Dysphagia Journal, which includes the scale on the 3rd page of the article.
Take Home Message: Secretions correlated more strongly with pneumonia than the PAS score. SLPs can be instrumental in raising awareness that excessive secretions alone are a risk factor for developing pneumonia. In other words, even if you have a safe swallow, you could still develop pneumonia in absence of prandial aspiration if you cannot protect your airway from excessive secretions.
This team had a poster at DRS2017 as well showing that the reliability was adequate among untrained SLPs, even those with little to no FEES experience. (Please see the citation below regarding the Hunting & Miles poster).
The discussion after this session questioned the pathophysiology of the excessive secretions and how to treat them. Can we gently dry up secretions without overdrying or other side-effects (See also my prior drug-induced dysphagia blog)? There was speculation that the tracheotomy tube and nasogastric feeding tube themselves could explain the increased secretions. (Note: Dr Steven Leder always specified the term “tracheotomy” versus “tracheostomy”.) Additionally, these tubes may desensitize the upper aerodigestive tract to the presence of secretions. Finally, this very acute population may have a reduced saliva swallowing frequency (see last year’s DRS Digest under section #4 for more on swallowing frequency).
During the presentation, Miles recommended the following articles regarding how aspiration of secretions can be a predictor for pneumonia:
3) Sensory Enhancement Strategy? Carbonation!
As far back as 1995, Logemann looked at the stimulation effects of a sour bolus. (See my 2015 pre-DRS blog on Remembering Our Roots & Jeri Logemann). Many studies since have looked into stimulation effects of taste, bolus volume, temperature and carbonation. Leisa Turkington, from Brisbane, Australia, addressed carbonation with her research: “The Impact of Carbonation on Neurogenic Dysphagia during VFSS (Videofluoroscopic SwallowStudy.com).”
Turkington reviewed the small amount of literature on the topic already. For examples:
She also noted helpful review articles:
- See Turkington, et al., 2016, for a “narrative synthesis of existing evidence.”
- See Steele & Miller, 2010 for a review article about sensory input pathways and mechanisms in swallowing (See the Open Access free article here.)
- See Cathy Lazarus’ historical perspective in the 2017 Dysphagia journal, called “History of the Use and Impact of Compensatory Strategies in Management of Swallowing Disorders.” (Springer link: https://link.springer.com/article/10.1007%2Fs00455-016-9779-6)
The effect of carbonation on adults with known aspiration and neurogenic dysphagia was tested in this study, using 40% barium mixed with E-Z-Gas II (a packet of effervescent granules by EZ EM was added to 100ml of 40% barium). The participants were tested with “natural sips” of non-carbonated liquids versus natural “discrete sips” and “consecutive sips” of carbonated liquid.
Discrete sips of carbonated liquids reduced aspiration in 41% of participants. Scores on the Videofluorscopic Dysphagia Scale (VDS) were better on the carbonated trials. They found statistically significant improvements in PAS scores with carbonated liquids versus non-carbonated liquids, but quite a few participants still aspirated with the carbonated liquid (meaning some people had no change in scores and some had worse PAS scores). Of note, the researchers used the “worst swallow” from each condition for their comparisons, rather than the less meaningful method of taking the patient’s mean of all his/her PAS scores on a given condition.
Future research may want to compare exactly the same sip sizes and methods for the non-carbonated and carbonated trials. The “natural” sip of the non-carbonated liquid may have been significantly larger than the “discrete” sip of carbonated. Natural sip sizes have been found to be as large as 12-16ml in adults. The effervescence itself may cue the person to take a smaller sip. There was no comparison of consecutive sips of non-carbonated liquid to the carbonated liquid. Some participants may have aspirated more so on carbonated liquid due to the added condition of consecutive sips.
Audience discussion wondered if these effervescent granules in barium really have the same chemosensory and gustatory stimulation as our favorite ice-cold, super sweet sodas. Turkington mentioned research by Dietsch, et al., 2014 regarding barium’s affects on taste intensity and palatability.
Take Home Message: Carbonation may be able to reduce aspiration in adults with neurogenic dysphagia; however, we still need to test it in a case by case fashion. Potentially, this is one more condition to add to your videofluoroscopic swallow study (VFSS) or fiberoptic endoscopic evaluation of swallowing (FEES), especially if the person may refuse thickened liquids.
4) Why Did You Choose That Swallowing Strategy or Exercise?
Sometimes we throw the whole toolbox at a patient without knowing why we are choosing a particular dysphagia management tool. New research using novel imaging techniques may help answer our why questions.
The Effortful Swallow was imaged by a 320-row area detector computed tomography (ADCT) in Japan. Inamoto, Yoko, along with his colleagues from Fujita Health University, Toyoaoke, Aichi, Japan used 3D-CT posterior imaging with healthy young volunteers while they swallowed 4ml of a nectar thick liquid in semi-reclined positions. (Note: Dr Jeffery Palmer, from the Department of Physical Medicine and Rehabilitation at John Hopkins University, Baltimore, was a co-author on this study. Palmer was our Dodd’s-Donner Lecturer at #DRS2017). Their oral presentation and abstract was titled: “Effortful Swallow and Its Affects on Pharyngeal Volume: Kinematic Analysis Using 3D Dynamic Computed Tomography”
Per their research, the effortful swallow caused the following timing and physiological changes:
- Prolonged velopharyngeal closure,
- Prolonged duration of hyoid displacement and decent,
- Prolonged laryngeal vestibule closure,
- Prolonged true vocal cord closure,
- Increased hyoid superior movement, but
- Reduced anterior hyoid displacement.
- Increased duration of the minimum pharyngeal volume (i.e., a maximum pharyngeal contraction will result in a residual minimum volume of 0ml).
However, there was NO difference in the:
- Duration or magnitude of UES opening.
Take Home Message: The Effortful Swallow may be effective in pharyngeal clearance and to reduce pharyngeal residue, by bringing the minimum pharyngeal volume down to 0 with potentially enhanced pharyngeal constriction. (However, during the presentation, Inamoto noted that this is done by “promoting posterior tongue base movement,” but tongue base retraction measures were not mentioned. They related the changes in hyoid movement to the “hypothesized enhancement of tongue base retraction.”)
In the future, they will expand testing in bolus volumes and in the healthy elderly and disease states.
5) You Know What They Say About Assuming:
What do we know, What do we assume; and What don’t we know?
Dysphagia research has been around for almost 40 years, and now the field has been asking tough philosophical questions at #ASHA16 and at #DRS2017. (See my post-ASHA16 blog). This branch of philosophy is called epistemology. I learned that this year from Mershen Pillay, a Speech-language Pathologist and professor from University of KwaZulu-Natal, Durban, South Africa and a www.IDDSI.org board member. Pillay, had a unique poster asking the epistemological question (what truths underlie our theories of knowledge production) and “are these relevant (ecologically valid) for research, evaluation and treatment?”
The following discussion reflects how our field is asking questions about itself. The field is raising awareness about what we may not know.
On Thursday afternoon, Phoebe Macrae, PhD lead a large panel discussion called: “Addressing the Gaps in Dysphagia Rehabilitation.” Julie Barkmeier-Kraemer, PhD, CCC-SLP started it off probing: “What We Know, What We Assume and What We Don’t Know.” David Paydarfar, MD, Georgia Malandraki, PhD, CCC-SLP, BCS-S and Jay Rosenbek, PhD, CCC-SLP, BC-NCD were also on the panel.
Georgia Malandraki, PhD, CCC-SLP, BCS-S summarized the session well. She noted that the presenters agreed that:
“We must define the underlyning physiology and pathophysiology before we try to attempt different treatments…. There are a lot of things we do that are not really based on the underlining physiology of the patients. We are trying different things. A lot of us develop strengthening and exercise training, but we need to be looking at who’s the best candidate for this treatment and who is not.”
Malandraki suggested that maybe we need to be talking about different “dysphagias,” just as Barkmeier-Kraemer reminded us of the different types of dysarthrias.
David Paydarfar, MD, from the University of Massachusetts Medical School, stressed how:
“All clinicians are neurologists, as the swallow is a microcosm of a larger universe.”
This certainly reminds us to consult with the neurology team. Malandraki noted that much research has focused on the brainstem, but that there is a lot to understand about the networking and communications across the brain, especially in how the brain adapts to lesions/impairments. Paydarfar introduced the concept that the basal ganglia is like the “ignition-break-clutch system” for all the central processing generators, “using higher order (cortical) command signals.” He called the cerebellum the “active steering mechanism,” which modulates the speed, effort and range of movement using feedback loops from motor and sensory input. We are looking forward to future dysphagia research into the basal ganglia, insula, cerebellum and more (see last year’s DRS Digest for a discussion of Dr Ianessa Humbert’s talk on the cerebellum).
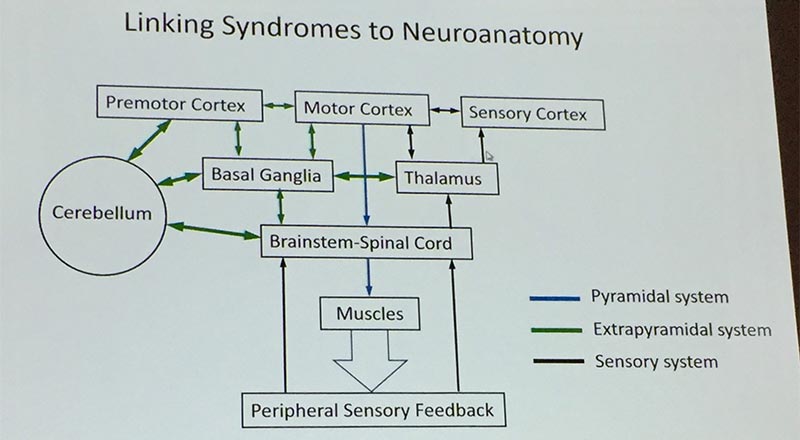
Dr David Paydarfar’s simplified neuroanatomy linkages. Modulations from basal ganglia and cerebellum. Fine tuning from the sensory feedback. He gave the example of all the cortical fine tuning that happens when we eat with proper etiquette!
Per Paydarfar, it is so important for clinicians to have accurate definitions, such as the following that came from his presentation:
- Paresis: Partial loss of muscle strength
- Paralysis: complete loss; same as plegia
- Hypotonia: reduced resistance to passive movement; floppy
- Rigidity: involuntary sustained muscle contraction
- Spasticity: increased resistance to passive strength with velocity of stretch, and hyperactive tendon reflexes
- Dystonia: intermittent, arrhythmic, brief or prolonged contractions of a muscle or group of muscles; termed myoclonus when very brief (100msec).
- Tremor: involuntary rhythmic movements produced by alternating contractions of reciprocally innervated muscles. Clonus is rhythmic contraction of a single myotome (
- Bradykinesia: slowness in the execution of a volitional movement (strength preserved).
- Hypokinesia: paucity of movement (strength preserved).
- Ataxia: incoordination of volitional movement due to abnormalities in the rate, range, and timing of motor sequences.
Take Home Message: “Not all dysphagia is weakness,” per Malandraki, and strengthening is not all it takes to remediate a swallow.
What if what we are doing causes maladaptive behaviors?
The problem is we only know what we can see on imaging (plus from observations, a thorough medical record review and case history from the patient/caregivers). When we see residue, we may assume that a paresis or hypotonicity are causing the weakness, launching our plan into strengthening exercises. David Paydarfar, MD noted how we cannot define hyper versus hypotonicity on a videofluoroscopic swallow study; we need to interact with the muscles (and seak out our neurologists).
Case Examples:
a) Phoebe Macrae presented a case of a patient of Maggie-Lee Huckabee’s who has spastic cerebral palsy and dysphagia causing significant pharyngeal residue. Rather than hypotonicity, this patient’s “weakness” came from spasticity or hypertonicity. There was “so much muscle, but so weak,” per Macrae. Additionally, there was decreased sensory input and range of motion. The swallow function was improved with use of sEMG biofeedback to train the patient to reduce the high tone in the submental muscles and coordinate a more functional swallow. This person needed retraining every 6-9 months for 5 years, but then the behavior lasted.
b) Another case was that of a person with compression from a tumor at the cerebellar-pontine angle. Potentially, the person had been trained in the past to use the effortful swallow. Using manometry, they saw his superior and inferior pharyngeal waves stacked on top of each other, firing together rather than in succession. He said he was “swallowing harder.” He ended up with this maladaptive behavior. He was able to use manometry as biofeedback to separate the superior pharyngeal constrictor activity from the inferior pharyngeal constrictor activity. Macrae noted that this feedback exercise enlists the bulbar-cortical loops. Once he was able to adjust the timing of the loop with daily practice for 2 weeks, he did not need to come back for further therapy. Granted, training with manometry in this way is invasive and expensive, but Malandraki noted that there is promise in targeting these sensory-motor loops with behavioral training and not just strengthening exercises.
Take Home Message: Interconnectedness of the loops – Training using sensory and biofeedback may have profound effects on motor output. Malandraki pointed to kinesiology research that has used sensory/proprioceptive training in the limbs of children with unilateral cerebral palsy to improve motor function. Similarly, Paydarfar described how if sensory feedback is severed, one’s muscles will become hypotonic and weak.
Some final wisdom by Jay Rosenbek, PhD, CCC-SLP, BC-NCD, from the University of Florida, Gainesville:
“We don’t know the active ingredients of many of our treatments.”
“We don’t know for sure the interaction of some treatment approach that we use and the influence on the swallow of hyper/hypotonicity, ataxia, apraxia and the rest.”
He urges us “not to go screaming into the night,” but rather to develop refined treatments based on “exquisite hypothesizing about (sensorimotor) control abnormalities.”
Thank you for reading. Please share your thoughts on these DRS Digest items and on other important #DRS2017 research. Thank you to all the researchers and attendees for your work in raising awareness of swallowing and swallowing disorders.
DRS’s multidisciplinary-global reach is unique! #DRS2017 had 199 new attendees out of the record-breaking meeting attendance of over 500. That growth had to be due in part to our social media presence. DRS is a growing and a vital part of advancing the science of swallowing and swallowing disorders. Please help in these efforts by continuing to share with your colleagues and connections on social media.
Citations of Scientific Papers & Posters:
Hunting, A. & Miles, A. (2017, March). The Development and Reliability of the New Zealand Secretion Score. Poster presented at the Dysphagia Research Society Annual Meeting, Portland, OR.
Ianmoto, Y., Saitoh, E, Itoh, Y, Aoyagi, Y., Shibata, S., Kagaya, H., et al. (2017, March). Effortful Swallow and its Affects on Pharyngeal Volume: Kinematic Analysis Using 3D Dynamic Computed Tomography. Scientific paper presented at the Dysphagia Research Society Annual Meeting, Portland, OR.
Lee, Y.S., Yap, X.Y., Chia, A.A. & Wong, S.M. (2017, March). Oral Health Screening by Speech and Lanugage Pathologists (SLPs) Using The Oral Health Assessment Tool (OHAT). Scientific paper presented at the Dysphagia Research Society Annual Meeting, Portland, OR.
Miles, A., McFarlane, M. & Hunting, A. (2017, March). Predictive Value of the New Zealand Secretion Scale (NZSS) for Pneumonia. Scientific paper presented at the Dysphagia Research Society Annual Meeting, Portland, OR.
Miles, A., Hunting, A., McFarlane, M. Caddy, D. & Scott, S. (2017). Predictive Value of the New Zealand Secretion Scale (NZSS) for Pneumonia. Dysphagia, 32, 1-8. DOI 10.1007/s00455-017-9841-z
Pillay, M. (2017, March). An Ecological-Epistemological Analysis of Dysphagia Research and Practice. Poster presented at the Dysphagia Research Society Annual Meeting, Portland, OR.
Turkington, L., Ward, E. & Farrell, A. (2017, March). The Impact of Carbonation on Neurogenic Dysphagia During VFSS (Videofluoroscopic SwallowStudy.com). Scientific paper presented at the Dysphagia Research Society Annual Meeting, Portland, OR.
Other articles cited in text:
Chalmers, J., Johnson, V., Tang, J.H., Titler, M.G. (2004). Evidence-based protocol: Oral hygiene care for functionally dependent and cognitively impaired older adults. Journal of Gerontological Nursing, 30(11), 5-12.
Kayser-Jones, J., Bird, W.F., Paul, S.M., Long, L. & Schell, E.S. (1995). An instrument to assess the oral health status of nursing home residents. Gerontologist, 35, 814-824.
Logemann, J.A., Pauloski, B.R., Colangelo, L., Lazarus, C., Fujiu, M. & Kahrilas, P.J. (1995). Effects of a sour bolus on oropharyngeal swallowing measures in patients with neurogenic dysphagia. J Speech Hear Res, 38(3), 556–63.
Ota, K., Saitoh, E., Baba, M. & Sonoda, S. (2011). The secretion severity rating scale: A potentially useful tool for management of acute-phase fasting stroke patients. J Stroke Cerebrovasc Dis, 20 (3), 183-187.doi: 10.1016/j.jstrokecerebrovasdis.2009.11.015.
Takahashi, N., Kikutani, T., Tamura, F., Groher, M. & Kuboki, T. (2012). Videoendoscopic assessment of swallowing function to predict the future incidence of pneumonia of the elderly. J Oral Rehabil, 39 (6), 429-437. doi: 10.1111/j.1365-2842.2011.02286.x.


